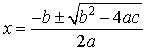CH223
- 11:00
EXAM-2B
Practice
|
Information: |
|
gas
constant: R = 0.08206 L·atm/mole·K
= 8.314 J/mole·K
Avogadro's number: NA
= 6.022 ´
1023/mol
Q-dependence of free energy change: DG
= DGo
+ RT ln(Q)
normal freezing point of water:
0°C = 273K
water ionization constant (25°C): Kw = 1.00 ´
10-14
pressure: 1 atm = 760 mmHg
= 760 torr
log conversion: ln(x) =
2.303 log(x)
quadratic equation:
ax2 + bx + c = 0, |
|
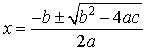 |
Multiple
choice: select
one answer for each question:
1.
Calculate
K for HCN(aq)
+ OH-(aq)
 CN-(aq)
+ H2O(l) from the reactions
CN-(aq)
+ H2O(l) from the reactions
|
|
HCN(aq)
 H+(aq)
+ CN-(aq)
, K = 4.9 ´10-10
H+(aq)
+ CN-(aq)
, K = 4.9 ´10-10
H2O(aq)
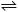 H+(aq)
+ OH-(aq)
,
K = 1.0 ´10-14
H+(aq)
+ OH-(aq)
,
K = 1.0 ´10-14 |
|
|
|
|
a) |
4.9 ´
104 |
|
b) |
2.0
´
104 |
|
c) |
4.9
´
10-24 |
|
d) |
2.0
´
10-5 |
|
e) |
2.0
´
10-25 |
|
|
|
2.
CaCO3 has Ksp=
4.8 ´10-9 .
The molar solubility of calcium carbonate in water is
|
|
|
|
a) |
4.8
´
10-9 M |
|
b) |
7.3
´
10-4 M |
|
c) |
6.9
´
10-5 M |
|
d) |
7.3
´
10-7 M |
|
e) |
6.7
´ 10-3 M |
|
|
|
3.
In which of the following reactions is DS°
negative?
|
|
1.
I2(g) ®
I2(s)
2. H2O(l)
®
H2O(s)
3. CO2(g)
+ CaO(s) ®
CaCO3(s) |
|
|
|
|
a) |
1
only |
|
b) |
2
only |
|
c) |
3 only |
|
d) |
1 and
2 only |
|
e) |
1, 2
and 3 |
|
|
|
4. Find
the quotient [Ag+ ]/[Ag(CN)2-
] from the complex ion
formation reaction
Ag+(aq)
+ 2 CN-(aq)
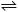 Ag(CN)2-
(aq), Kf = 3 ´1020
Ag(CN)2-
(aq), Kf = 3 ´1020
when the equilibrium concentration of CN-
is 0.10 M.
|
|
|
|
|
a) |
3 ´
10-19 |
|
|
b) |
3 ´
10-4 |
|
|
c) |
3 ´
10+2 |
|
|
d) |
3 ´
10-6 |
|
|
e) |
3 ´
10-22 |
|
|
|
|
|
5. The pH after mixing
0.500000 mole of HCl and 0.499996 mole of NaOH in 1000 mL of water is:
|
a) |
5.88 |
|
b) |
5.13 |
|
c) |
5.40 |
|
d) |
4.92 |
|
e) |
5.28 |
|
|
|
6. Spontaneous
chemical reactions at constant T and P always have __________.
1. DS
> 0 ,
2. DH
< 0,
3. DG
< 0
|
|
|
|
a) |
1 only |
|
b) |
3
only |
|
c) |
1 and
2 only |
|
d) |
2 and
3 only |
|
e) |
1, 2
and 3 |
|
|
|
7.
The pH of a solution that contains 4.8
mole of benzoic acid C6H5COOH (pKa = 4.20) and
3.6 mole of sodium benzoate C6H5COONa in 927 mL of water
is:
|
|
|
|
|
a) |
3.91
|
|
b) |
4.32 |
|
c) |
4.15 |
|
d) |
4.08 |
|
e) |
4.24 |
|
|
|
8.
When DSo
< 0 and DHo
< 0 a chemical reaction favors
|
|
|
|
|
a) |
products
at low temperature and products at high temperature |
|
b) |
products
at low temperature and reactants at high temperature
|
|
c) |
reactants
at low temperature and products at high temperature |
|
d) |
reactants
at low temperature and reactants at high temperature |
|
e) |
none of the above |
|
|
|
9.
Calculate
the equilibrium constant for the reaction at 298K:
|
|
|
3C(s)
+ 4H2(g)
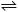 C3H8(g),
DH°
= -103.8 kJ, DS°
= -269 J/K C3H8(g),
DH°
= -103.8 kJ, DS°
= -269 J/K |
|
|
|
|
a) |
1.0
´
1017 |
|
b) |
2.1
´
1032 |
|
c) |
1.0 |
|
d) |
3.7
´
1019 |
|
e) |
1.4
´
104 |
|
|
|
10.
Calculate
the solubility of Cd(OH)2 (Ksp =1.2 ´10-14)
in a solution buffered at pH = 7.00.
|
|
|
|
a) |
0.3 M
|
|
b) |
4.8 M |
|
c) |
0.6 M |
|
d) |
1.2 M
|
|
e) |
5.0 ´10-8
M |
|
|
|
| Questions 11-15:
Titrate 72 mL of 0.200M formic acid HCOOH (Ka = 1.80 ´
10-4) with 0.200M NaOH. |
11.
The pH at the starting point is
|
|
|
|
a) |
1.80 |
|
b) |
3.42 |
|
c) |
2.85 |
|
d) |
3.16 |
|
e) |
2.22
|
|
|
|
12. The
volume of NaOH needed to just reach the buffer point is:
|
|
|
|
a) |
90
mL |
|
b) |
54
mL |
|
c) |
120
mL |
|
d) |
36
mL |
|
e) |
108 mL |
|
|
|
13.
The pH at the buffer point is
|
|
|
|
|
a) |
3.74 |
|
b) |
4.36 |
|
c) |
4.68 |
|
d) |
7.00 |
|
e) |
9.32 |
|
|
|
14. At
pH = 5.50 the quotient [HCOO-
] / [HCOOH] is:
|
|
|
|
a) |
36 |
|
b) |
18 |
|
c) |
57
|
|
d) |
69 |
|
e) |
180 |
|
|
|
15.
The pH at the equivalence point
is
|
|
|
|
|
a) |
7.00 |
|
|
b) |
8.37 |
|
|
c) |
7.94 |
|
|
d) |
8.62 |
|
|
e) |
9.43 |
|
|
|
|
|
| Questions 16-20: Titrate
84 mL of 0.125 M acid H2A (Ka1 = 3.7 ´
10-5 , Ka2 = 8.2 ´
10-10 ) with 0.125 M NaOH . |
16. The
volume of NaOH needed to just reach the first equivalence point is:
|
|
|
|
a) |
42
mL |
|
b) |
84
mL |
|
c) |
126
mL |
|
d) |
105
mL |
|
e) |
168
mL |
|
|
|
17.
The pH at the
first equivalence point is
|
|
|
|
|
a) |
8.27 |
|
b) |
7.00 |
|
c) |
4.43 |
|
d) |
6.76 |
|
e) |
8.14 |
|
|
|
18.
The
order of concentrations at the first equivalence point is:
|
|
|
|
a) |
[HA-
]
> [A2-
]
> [H2A] |
|
b) |
[H2A]
= [HA-
]
> [A2-
] |
|
c) |
[HA-
]
> [H2A] > [A2-
] |
|
d) |
[A2-
]
= [HA-
]
> [H2A] |
|
e) |
[HA-
]
> [A2-
]
= [H2A] |
|
|
|
19.
The order of
concentrations at pH = 7.00 is:
|
|
|
|
a) |
[HA-
]
> [A2-
]
= [H2A] |
|
b) |
[A2-
]
= [HA-
]
> [H2A] |
|
c) |
[HA-
]
> [H2A] > [A2-
] |
|
d) |
[H2A]
= [HA-
]
> [A2-
] |
|
e) |
[HA-
]
> [A2-
]
> [H2A]
|
|
|
|
20.
The order of concentrations at
the second equivalence point is:
|
|
|
|
a) |
[HA-
]
> [H2A] > [A2-
] |
|
b) |
[A2-
]
= [HA-
]
> [H2A] |
|
c) |
[A2-
]
> [HA-
]
> [H2A] |
|
d) |
[HA-
]
> [A2-
]
= [H2A] |
|
e) |
[H2A]
= [HA-
]
> [A2-
]
|
|
|
|

| KEY: |
 |